 .png)
On the other hand, we do know how to write down the Hamiltonian operator for nearly any conceivable system. Such an equation is known as the Schröedinger equation:įor many systems, including multi-electron atoms and molecules, the Schrödinger equation is difficult to solve because we do not know beforehand what the wavefunction of a complex system such as a molecule looks like. 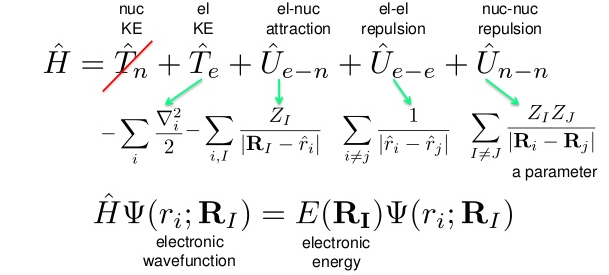
For example, the energy of a molecule can be obtained by solving the equation in which the Hamiltonian operator acts on the wavefunction. 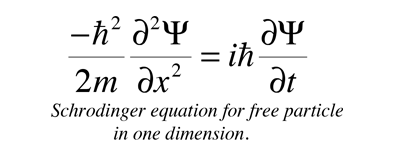
The properties of the system are obtained by acting on the wavefunction with a property-specific operator. In quantum mechanics, a system is characterized by its wavefunction Ψ. Semiempirical Quantum Chemistry: Introduction.īrief Introduction to Quantum Mechanics Schrödinger Equation 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
